Research projects
Our state-of-the-art laboratory at Woods Hole Oceanographic Institution focuses on marine organisms that grow shells in the ocean, and their relationship to the marine carbon and alkalinity cycles. We conduct experiments and make measurements in the lab and in the field, using a combination of novel instrumentation and innovative approaches to traditional carbonate chemistry measurements.
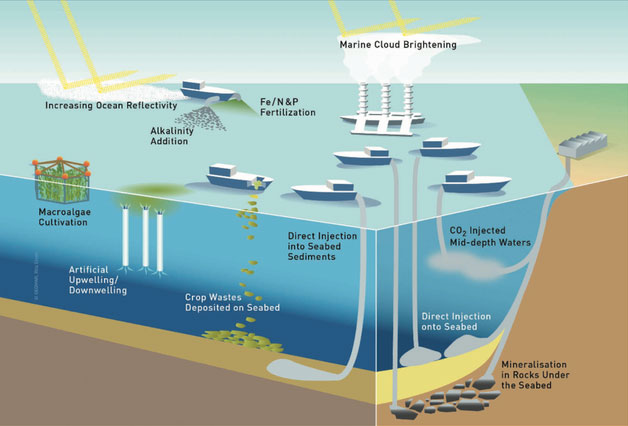
Impacts and biological feedbacks associated with ocean alkalinity enhancement
Ocean alkalinity enhancement (OAE) is an approach to mitigating anthropogenic CO2 emissions that involves increasing the already substantial buffering capacity of the world oceans. I am the lead PI of the Locking Ocean Carbon in the Northeast Shelf and Slope (LOC-NESS) Project, where we are addressing the effectiveness and environmental impacts of OAE. Check out more of our research on this topic at our project website.
We also study natural analogs to OAE, including alkalinity delivery to seawater, specifically in the Mississippi River and its plume in the Gulf of Mexico.
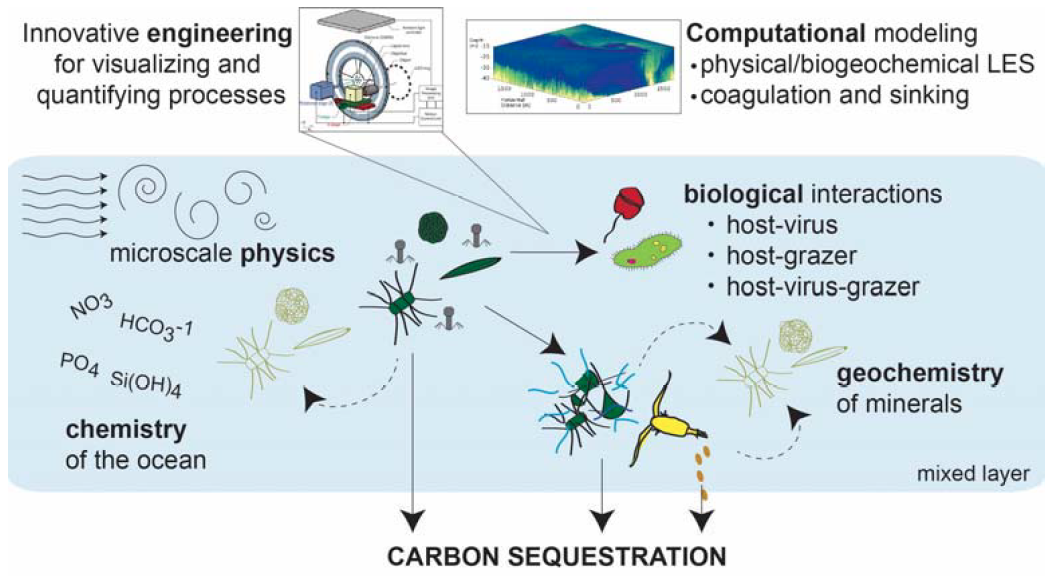
Carbon and alkalinity cycling driven by coccolithophores, zooplankton, and marine viruses
The ocean carbon cycle is constantly in flux, with organisms growing in the surface ocean, dying, being eaten, and sinking down to the depths where carbon is stored away from the atmosphere. It turns out that marine viruses are a key part of the life cycle of coccolithophores, and we are investigating the linkages between coccolithophores, zooplankton grazing, and viral infection. This project is funded through NSF's Growing Convergence Research (GCR) program, led by PIs Kay Bidle and Kim Thamatrakoln. Aspects of this project include:
- Grazing experiments feeding isotopically labeled coccolithophores to microzooplankton, to determine how much CaCO3 dissolution happens during this feeding process (JP student Chloe Dean)
- Developing a 13C-based assay for calcification rates in the laboratory and in the field (C. Dean)
- Dissolution driven by the respiration of organic matter in marine snow aggregates (B. Borer and A. Babbin, MIT)
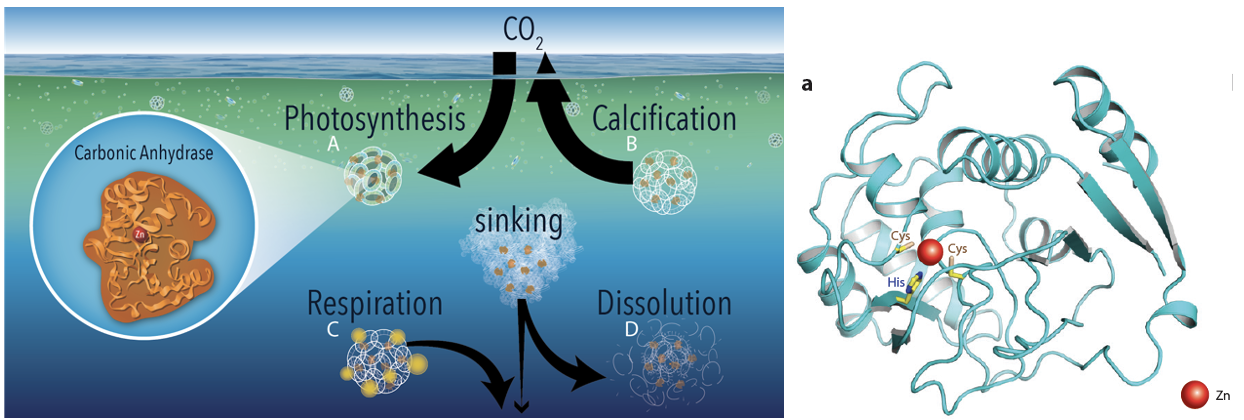
Marine Carbonic Anhydrase Biochemistry
In this project with Co-PI Mak Saito (WHOI), we are unpacking the activities of diatom-based carbonic anhydrases by pairing sensitive assays of enzyme activity with state-of-the art proteomic analyses. Aspects of the project include:
- Laboratory studies of CA expression as a function of zinc and pCO2 concentrations paired with proteomics (Emily Burdige, JP Student)
- Field surveys of CA expression across productivity gradients in the equatorial Pacific (E. Burdige)
- Metalloproteomics of marine diatoms (planned)
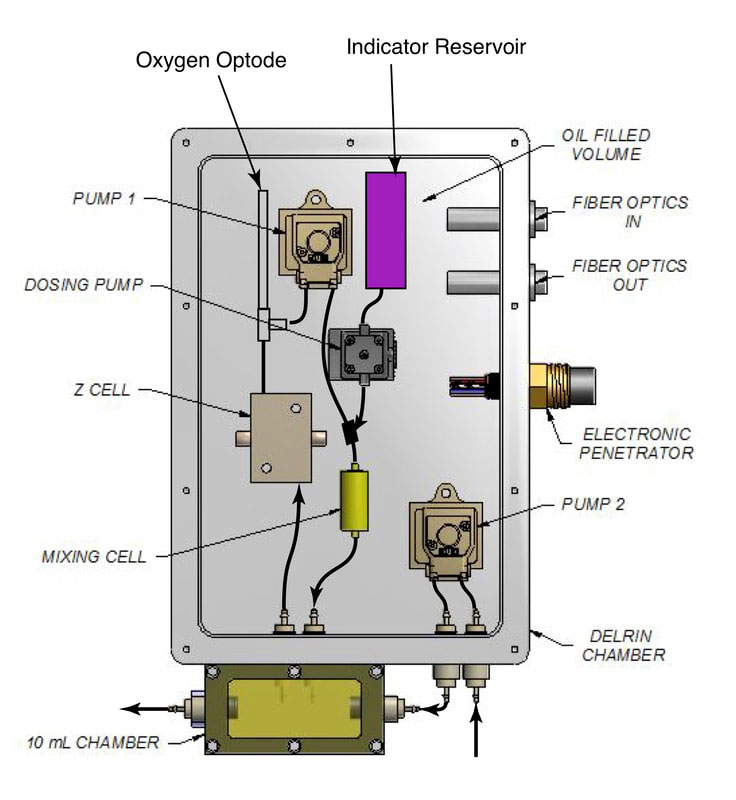
Biogenic calcium carbonate solubilities through lab and field saturometry
We do not fully understand the thermodynamic controls on calcite solution in the ocean. For example, the pressure dependence of calcite solubility has never been successfully reproduced in the field. The saturometer evaluates the reactivity and solubility of biogenic calcites and aragonites in the lab and the field across a range of heretofore underexplored thermodynamic variables. This device is being redesigned with a spectrophotometric pH sensor. This research is the focus of a funded NSF Chemical Oceanography proposal with co-P.I. Matt Long at WHOI.
Underwater Gradient Exchange Mass Spectrometry
Led by PI Matt Long (WHOI), we are building and underwater mass spectrometer paired with a novel "gradient exchange" seawater sampling system, that will allow us to simultaneously determine benthic fluxes of oxygen, carbon dioxide, sulfide, and methane.